Impact of BLC with Cysview
Blue Light Cystoscopy with Cysview® is clinically proven to detect NMIBC missed by White Light Cystoscopy alone.1
Confidence at First Sight
Blue Light Cystoscopy (BLC®) with Cysview is FDA-approved for use in both transurethral resection of bladder tumor (TURBT) and surveillance. By having better visibility and detection during initial examinations and resections, urologists can improve diagnosis and management of non-muscle invasive bladder cancer (NMIBC).1
Because Cysview causes cancer cells to glow bright pink under blue light, tumor margins and hard-to-see lesions become more clearly visible during a cystoscopy. As a result, the cancer can be more thoroughly removed by TURBT.
A complete TURBT is critical in NMIBC management for accurate tumor typing, staging, and grading, as well as optimization of patient outcomes. An incomplete TURBT can lead to under-staging, misdiagnosis and incomplete resection; and over-reliance on intravesical therapy may lead to less optimal treatment, worse outcomes and increased costs for patients.
TURBT procedures with BLC with Cysview have been shown to increase diagnostic accuracy and optimize patient care management decisions.1
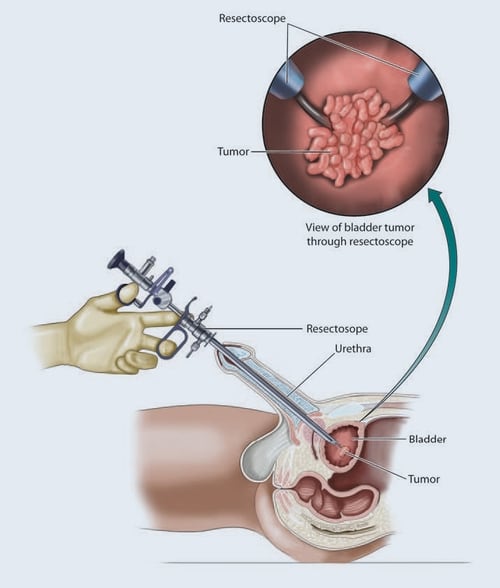
Reproduced from Bladder Cancer: A Patient-Friendly Guide to Understanding Your Diagnosis and Treatment Options. Copyright© 2017 David Pulver
Cysview is approved for use with:

TURBT
The 2016 AUA/SUO NMIBC Guideline recommends: In a patient with non-muscle invasive bladder cancer (NMIBC), a clinician should offer blue light cystoscopy at the time of TURBT, if available, to increase detection and decrease recurrence. (Moderate Recommendation; Evidence Strength: Grade B)2
BLC with Cysview offers optimal diagnosis right from the first TURBT.
Bladder Cancer is difficult to manage
- When TURBT is performed with only white light, up to 75% of patients present with tumors at repeat TURBT3
BLC with Cysview leads to improved disease management
- Approved for use in OR and office1
- Detects more tumors than white light alone1
- Better detection may lead to appropriate risk categorization and improved patient management and outcomes4
Surveillance
In surveillance, Blue Light Cystoscopy with Cysview can be performed in the office or clinic for follow-up cystoscopies. In the phase III study that resulted in the expanded Cysview indication to include patients undergoing surveillance cystoscopy for carcinoma of the bladder, results showed that Cysview significantly (p<0.0001) improves detection of patients with recurrent bladder cancer.1
The detection of additional tumors in the surveillance setting may drive earlier diagnosis of high-risk lesions that would have otherwise been missed with white light alone. Improved detection during surveillance cystoscopies can have a profound effect on disease management in intermediate and high-risk bladder cancer patients.1
Experts recommend using BLC with Cysview for surveillance. The more confidence a urologist can have in the impact of disease management, the more appropriate the disease management efforts can be.4
Patients Like Cysview
Patients seem to agree. In a prospective, multi-center study, patients reported that BLC with Cysview in the surveillance setting was worthwhile and would recommend it to others.5
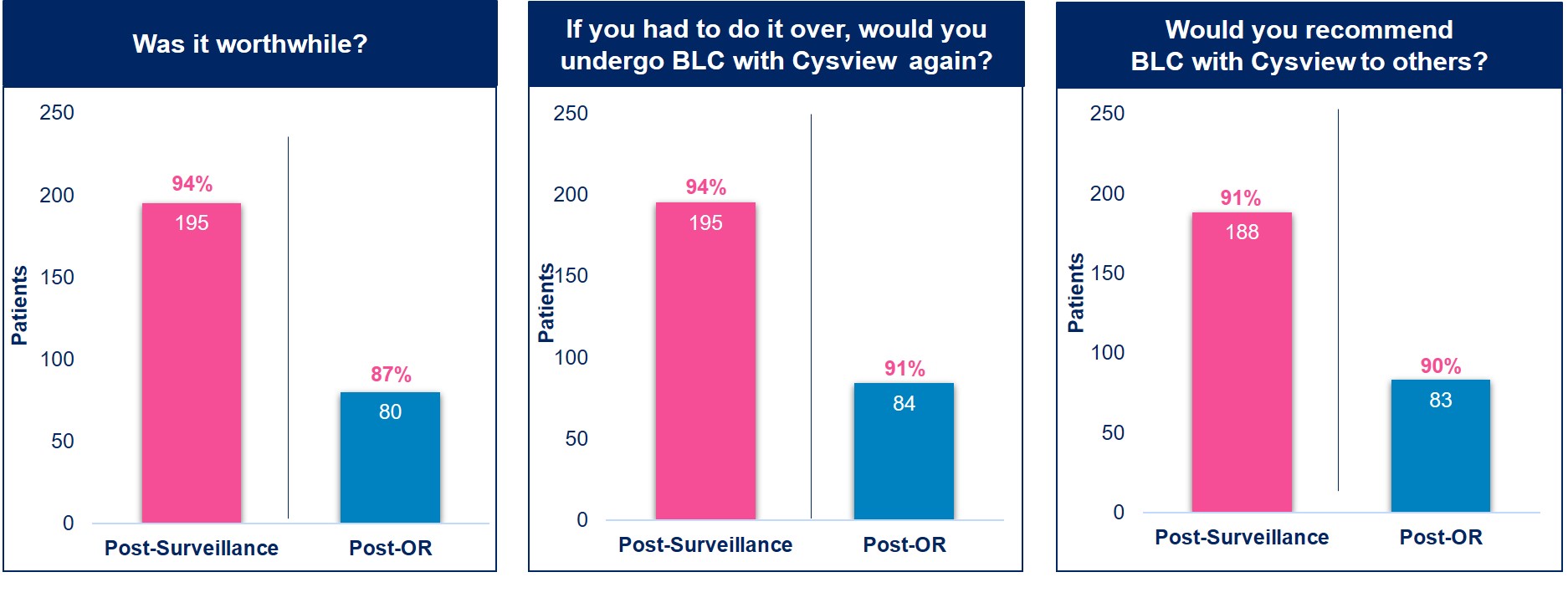
The majority of patients thought Blue Light Cystoscopy with Cysview was worthwhile and would recommend it to others.
1 Cysview [prescribing Information]. 2019:1-4.
2 Chang SS, Boorjian SA, Chou R, et al. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline. J Urol. 2016;196(4):1021-1029.
3 Cumberbatch MGK, Foerster B, Catto JWF, et al. Repeat Transurethral Resection in Non-Muscle Invasive Bladder Cancer: A Systematic Review. Eur Urol. 2018;73(6):925-933.
4 Richards KA, Smith ND, Steinberg GD. The Importance of Transurethral Resection of Bladder Tumor in the Management of Nonmuscle Invasive Bladder Cancer: A Systematic Review of Novel Technologies. J Urol. 2014;191(6):1655-1664.
5 Smith AB, Daneshmand S, Patel S, et al. Patient-Reported Outcomes of Blue-Light Flexible Cystoscopy with Hexaminolevulinate in the Surveillance of Bladder Cancer: Results from a Prospective Multicentre Study. BJU Int. 2019; 123(1):35-41.


